There are alternatives like nickel and manganese, but these can have stability issues; over time, nickel and manganese can dissolve in the electrolyte and damage the battery system.
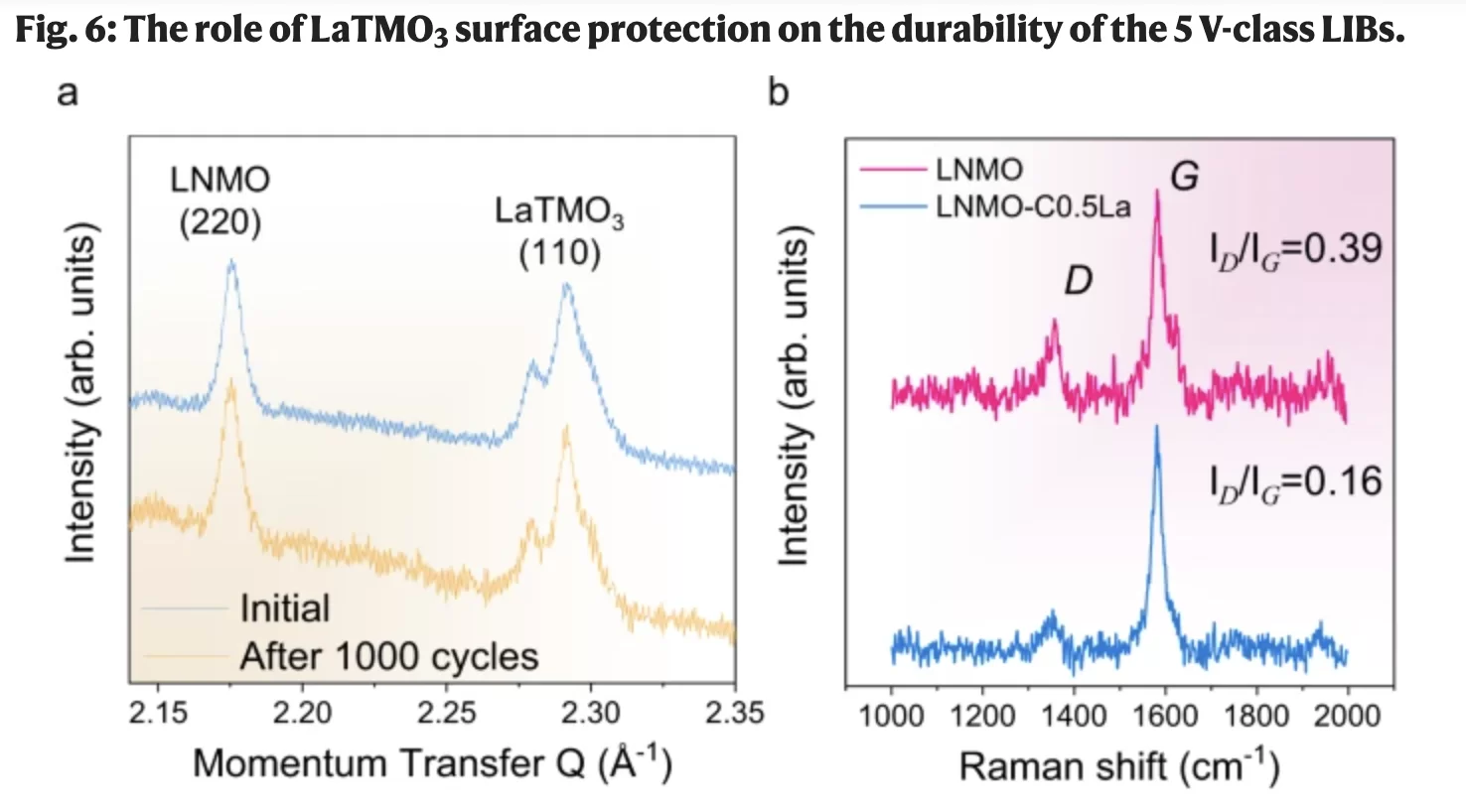
Working with a lithium-nickel-manganese cathode material, Wang and his team developed a coating using a technique called epitaxy. It works by growing a single atom thick layer of crystal, made up of lanthanum, nickel, manganese and oxygen, on the cathode particles.
This protective layer enables a high voltage battery – approximately 4.5V – and helps reduce cathode corrosion.
“This is the key innovation of the work, with this single-layer protection, the dissolution of nickel and manganese becomes more difficult,” Wang said.
In a test facility, the team ran batteries for a few months at a time to monitor performance and determined approximately 77% capacity retention after 1000 cycles when tested in combination with an anode graphitic carbon and a non-aqueous electrolyte solution.
Scaling and Commercialization
Wang said the shielding material could be on the market in two to three years – but there’s still work to be done. The next step is to scale up – moving from coin-sized battery testing to medium and larger scale – in partnership with industry and CSIRO.
A recently launched $242.7 million federal government program, Pioneerhopes to accelerate the commercialization of research like this.
It was announced in April that Curtin University will lead the Critical Resources and Minerals component of the program, in partnership with UQ and James Cook University.
Rohan McDougall, Chief Marketing Officer at Curtin, said create 33 industrial partners are on board.
“They range from companies looking to process and add value to minerals, [such as] Linus who plan to work together to optimize rare earth processing in Australia and their processing facilities internationally,” he said.
“There are others looking at products ranging from processing lithium, to improving efficiency and mining and processing elements like nickel and cobalt, and even producing cathodes and anodes and battery cells.”

